|
12/10/2023 0 Comments Macrophages in synovial fluid
OSM differentially regulates pro-inflammatory mechanisms, significantly promotes pro-angiogenic and invasive mechanisms and alters cellular bioenergetics in favour of glycolysis in RAFLS and HUVEC. Finally, CD206+CD163+ macrophages induce enhanced autologous T cell responses and spontaneously secrete high levels of IL-8, IL-6 and TNFa.įinally, the effect of key macrophage-secreted cytokines, OSM and TNFa on stromal cell (HUVEC/RAFLS) function was examined. Moreover, we demonstrate differing metabolic demands between CD206+CD163+ and CD206-CD163- macrophage subsets using FLIM analysis. We reveal that the CD206+CD163+ macrophage population is transcriptionally distinct from synovial fluid, double negative CD206-CD163- and pure monocyte-derived M1/M2 macrophages, with unique tissue-resident gene signatures. CD206+CD163+ and CD206-CD163- macrophage populations were sorted from RA synovial tissue and synovial fluid and RNA-seq analysis performed. Furthermore, the CD206+CD163+ macrophage subset is present in healthy synovial tissue but does not express CD40. We demonstrate that this subset is enriched in synovial tissue compared to fluid but specifically in RA synovium compared to OA and PsA synovial pathotypes. Within this spectrum we identify for the first time, that the dominant macrophage population residing in the RA synovium is a transitional subset of tissue-resident CD206+CD163+ macrophages that display elevated CD40 expression. In this thesis we phenotypically characterise RA synovial tissue macrophages revealing a spectrum of macrophage activation states that don?t conform to the binary M1/M2 framework in vivo. Mechanistically, we demonstrate that the hyper-inflammatory and metabolic phenotype of RA monocytes and M1 macrophages, is mediated through STAT3 phosphorylation whereby inhibition of STAT3 activity switches the pathogenic phenotype of myeloid cells for resolution of inflammation.ĭistinct subsets of tissue macrophage in the inflamed RA synovium remain largely unexplored. Marked transcriptional variance was indicated by RNA-seq, with a key role for STAT3 activation in macrophage polarisation identified. Furthermore, analysis of polarised RA M1/M2 macrophages reveals divergent inflammatory, bioenergetic and phagocytic functions. In addition, we demonstrate a consistent upregulation of glycolytic machinery indicating fundamental abnormalities in glucose processing in RA myeloid cells. 
RA M1 macrophages replicate the inflammatory memory bias of their precursor cells demonstrating heightened glycolysis and mitochondrial respiration coupled with altered mitochondrial morphology compared to HC. Interestingly, the hyper-inflammatory, hyper-metabolic phenotype of RA monocytes persists following differentiation into ex vivo macrophages. Metabolic analysis of RA monocytes using the Seahorse Flux analyser reveals a robust boost in both OXPHOS and glycolysis in RA CD14+ monocytes compared to HC. In this study we demonstrate that circulating RA CD14+ monocytes are primed to produce pro-inflammatory mediators, a phenotype indicative of M1-like macrophage polarisation. Therefore, the aims of this thesis are to elucidate the distinct pathogenic functions, bioenergetic demands and activation status of monocytes, monocyte-derived macrophages and synovial tissue macrophage subsets in RA. Despite this, the precise nature and function of infiltrating monocyte-derived macrophages and their precursor cells are poorly defined in RA with even less known about RA synovial tissue macrophages. However, it is now known that macrophages are heterogeneous both in function and origin with many macrophages seeded during embryonic development independently. Upon entry into the synovium, peripheral blood monocytes differentiate into the dichotomous M1 (pro-inflammatory) or M2 (anti-inflammatory) macrophage activation states. Macrophages are an exquisitely plastic pool of innate cells critically involved in directing the immune response in RA. 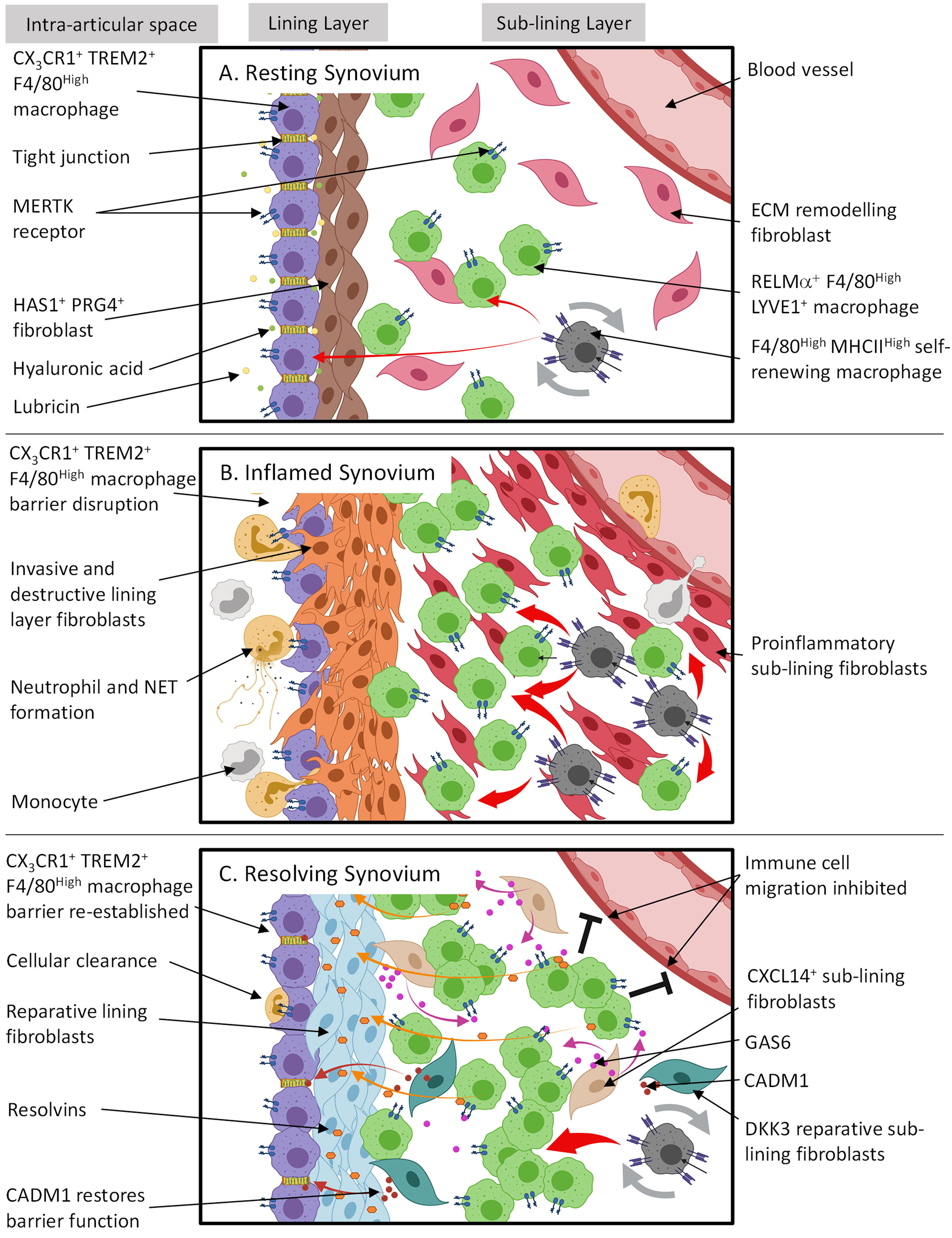
Hanlon, Megan, Macrophages are critical drivers of synovial inflammation in Rheumatoid Arthritis, Trinity College Dublin, School of Medicine, 2020
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
